CoolStat – a completely new way to manage patient temperature.
Designed to provide effective cooling that is simple, safe, and easy-to-use.

COOLSTAT
CoolStat is a completely new way to manage patient temperature. Unlike other cooling systems, CoolStat does not use cumbersome cooling pads or large refrigeration carts. CoolStat is the ultimate in simplicity. It cools using only dry, room temperature air, based on a simple technique referred to as evaporative cooling.
SCIENCE APPLIED TO COOLING
HOW IT WORKS
CoolStat blows dry, room temperature air into one nostril in a unidirectional fashion before flowing freely out the other nostril and the mouth. The nasal turbinates are highly vascularized, mucus-containing membranes with a large, convoluted surface area. As the dry air from CoolStat flows over the moist turbinates, it induces the liquid water and mucus on the turbinates to change phase, from a liquid to a gas. This phase change pulls energy out of the body, causing systemic cooling.

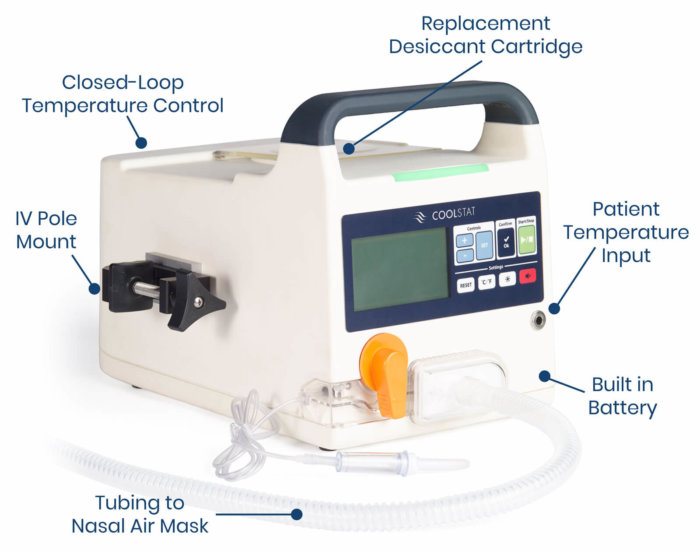
COOLSTAT FEATURES
Easy to install and operate
Only portable device - cooling anywhere!
Connects seamlessly to hospital monitor/EMR
Designed to be comfortable for patient
Designed to provide clinically effective cooling
Designed to reduce or eliminate shivering response
Very little infection risk (non-invasive)
Lower cost than other targeted temperature management (TTM) systems
FAQ SECTION
How is CoolStat Used?
The CoolStat device is small, lightweight and portable, and can move throughout the hospital with the patient. The device generates a source of filtered air that is delivered to the patient via a single-use air tubing set. The only interface with the patient is a simple nasal mask. The CoolStat includes a closed-loop temperature control system. That means that the operator of the device can select a target temperature, and the CoolStat is designed to automatically cool and hold the patient at the target temperature.
What indications is the CoolStat designed to treat?
CoolStat has not yet been cleared or approved for use. It is currently designated for investigational use only. The device is currently being used in clinical studies in neurogenic fever (central fever) that can develop following a stroke, traumatic brain injury, seizure and metabolic encephalopathy. The first multi-center clinical study using CoolStat was completed in 2022. We are also exploring the use of CoolStat to initiate mild hypothermia in a pre-hospital setting, post sudden cardiac arrest, and we have seen positive early results using transnasal cooling to reduce or eliminate the pain from migraine.
PUBLICATIONS
Rapid Induction of Therapeutic Hypothermia
Using Transnasal High Flow Dry Air
Efficacy and Safety of Transnasal CoolStat Cooling
Device to Induce and Maintain Hypothermia
Safety and Feasibility of a Novel Transnasal Cooling Device to Induce Normothermia in Febrile Cerebrovascular Patients.
Minimizing Shivering During Targeted Normothermia: Comparison Between Novel Transnasal and Surface Temperature-Modulating Devices
Contact Us

Mi-Helper, Inc. collaborates with distinguished scientists and clinicians on a mission to develop cutting-edge therapies that cater to the unmet needs in pain management and mood disorders.
*This product has not yet been cleared or approved for use. It is currently designated for investigational use only.
